The metaphase spindle is a nonequilibrium, steady-state ensemble primarily composed of microtubules that continually grow, shrink, and slide relative to each other, even as the spindle keeps a constant length. Despite the detailed molecular information that has been gained, it is unclear how the size and shape of the spindle results from the activities of the molecules from which it is compose. We are using a combination of quantitative experiments and theory to understand the collective behaviors of microtubules in the spindle: what they are, how they arise from mescosopic and molecular properties, and how they result in spindle assembly, morphology, and dynamics.

Prediction of director orientation in the spindle (Left) and corresponding measurement (Right) from averaging ∼5 min of an LC-Polscope movie (100 frames) of a spindle.
Contractile Systems:
Many cellular processes are driven by cytoskeletal assemblies. Cytoskeletal filaments and motor proteins organize into cellular scale structures and the molecular properties of cytoskeletal components can affect the large scale behaviors of these systems. We investigate the self-organization of stabilized microtubules in Xenopus oocyte extracts and find that macroscopic networks of microtubules spontaneously contract.
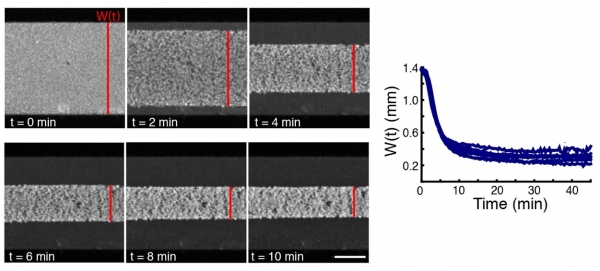
We propose that these contractions are driven by the clustering of microtubule minus ends by dynein. Based on this observation, and working with Michael Shelly's group, we have constructed an active fluid theory of network contractions which predicts a dependence of the timescale of contraction on initial network geometry, a development of density inhomogeneities during contraction, a constant final network density, and a strong influence of dynein inhibition on the rate of contraction. These results demonstrate that the motor-driven clustering of filament ends is a generic mechanism leading to contraction.

Extensile Systems:
-----
Publications:
. The Material Basis of Life. Trends in cell biology. 2015.![]() Download Paper
Download Paper
. Determining physical principles of subcellular organization. Developmental cell. 2014;29 :135–138.![]() Download Paper
Download Paper
. Physical basis of spindle self-organization. Proceedings of the National Academy of Sciences. 2014;111 :18496–18500.![]() Download Paper
Download Paper
. Nucleation and transport organize microtubules in metaphase spindles. Cell. 2012;149 :554–564.![]() Download Paper
Download Paper
. Nonequilibrium fluctuations in metaphase spindles: Polarized light microscopy, image registration, and correlation functions, in OPTO. International Society for Optics and Photonics ; 2010 :76180L–76180L.![]() Download Paper
Download Paper
-----
Project Members:
 |
Peter Foster Graduate Student Applied Physics Department |
 |
Postdoctoral Fellow Mathematics Department (NYU) |
 |
Bezia Laderman Graduate Student Physics Department |